While most of these programs are offered as majors within a specific academic department, in some cases subjects transcend departmental lines or are emphases within a major program. Of General Interest University Resources Undergraduate Studies Graduate Study and Research Descriptions of Undergraduates and Graduates Courses An Overview from Past and Present Administration, Faculty and Staff 3 15 23 41 Admission (610) 758-3100 College of Arts and Sciences (610) 758-3300 College of Business and Economics (610) 758-3400 College of Education (610) 758-3225 College of Engineering and Applied Science (610) 758-4025 69 261 271 Academic Calendar - inside back cover Undergraduate Major Programs The university offers the following undergraduate major programs. 
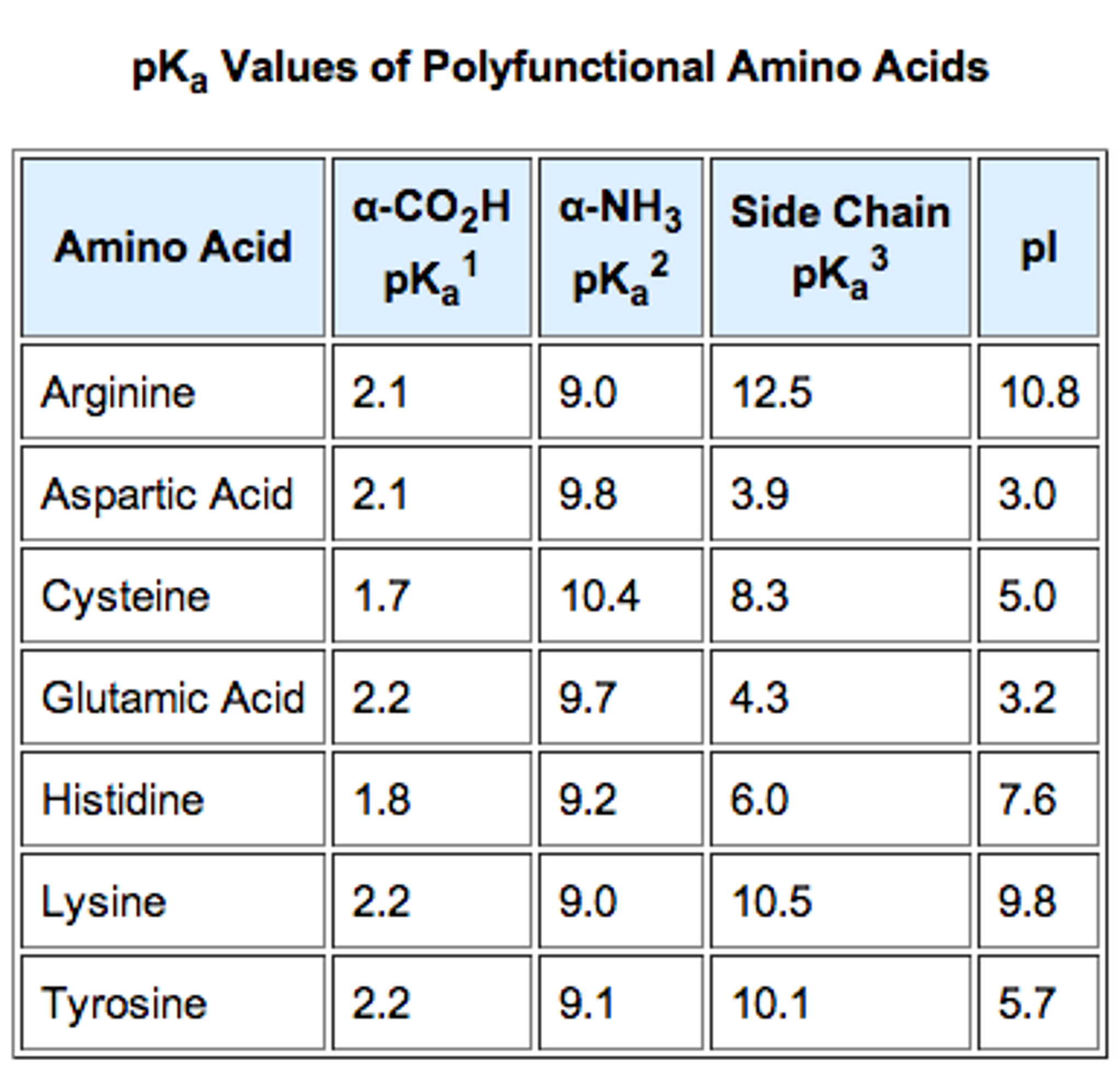
LEHIGH UNIVERSITY 1997-1998 COURSE CATALOG CONTENTS I. If you accidentally average all three pKas, you'll be able to spot your mistake because you'll start getting answers closer to 5 or 6 - which more closely resemble the pI of a neutral AA.Published since 1866 continuously, Lehigh University course catalogs contain academic announcements, course descriptions, register of names of the instructors and administrators information on buildings and grounds, and Lehigh history. Like said above, the actual pI of glutamic acid is a little bit greater than 3 - in other words, it's pretty low due to the acidity of the side chain. If you do this, you'll get an answer that is way too moderate. The most commonly made mistake by far happens when people average all three pKas for an acidic or basic AA. For basic AAs, like lysine, average the two HIGHEST pKas (which correspond to the amino terminal and the side chain). For acidic AAs, like glutamic and aspartic acid, average the two LOWEST pKas (which correspond to the carboxy terminal and the side chain). Acidic and basic amino acids, however, will have THREE pKas. For neutral amino acids like glycine, average the two pKas. Yes, this is a good answer! The easy way to think of this concept is, first, to figure out whether your AA is acidic, basic, or neutral (you should DEFINITELY have this memorized for the new MCAT). Let me know if you have any other questions Glutamic acid's real pI is listed as 3.08. 
The pKa of glutamic acid's side chain is around 4, so (4+2)/2 = 3. Thus we'll need to find the point where the carboxyls are both "half negative". In actuality, the pI for lysine is listed as 9.74, but it's enough to get you into the ballpark.Īs a further example, at a very low pH the alpha carboxyl and side chain carboxyl of glutamic acid will both be neutral while the alpha amino is positive. We can find this by averaging the pKas of the aminos: (9+10)/2 = 9.5. The isoelectric point is where the molecule is neutral, so we need to find the point where the aminos each equal "half of a positive charge" to balance out the negative carboxyl. At a high pH, such as 10-11, we know that the alpha amino group and the side chain amino group will both be neutral while the alpha carboxyl is negative. So then we can think about the amino acid at different pH values. For example, lysine's side chain pKa is around 10. The tricky part is when you calculate the pI of amino acids with electrically charged side chains. Their pI's will be in the 5.5 neighborhood, as the pKa of the amino group is roughly 9 and the pKa of the carboxyl is roughly 2. For those amino acids with side chains that are not electrically charged (which we can consider not ionizable), calculating the pI is as simple as averaging the pKa's of the alpha amino and carboxyl groups. This is my understanding, and anyone is free to correct me:Īll amino acids are ionizable, due to the alpha amino group and the alpha carboxyl group.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
