
Phosphorus is used to construct the basic building blocks of life, such as carbohydrates, lipids, and nucleic acids. It is also used to construct ATP and GTP. Oxygen is used to construct the basic building blocks of life, such as carbohydrates, lipids, and nucleic acids. Nitrogen is used to construct the basic building blocks of life, such as amino acids, nucleic acids, and proteins. Hydrogen is also used to construct ATP and GTP. The backbone rotation caused by the 2.78 5-Se-Me and 5-S-Me modications is due to the alteration of the water networking surrounding the uracil 5-position, which in turn changes the water interactions in the major groove and with the phosphate backbone. The helix is right-handed with 10 bp per turn. Frank-Kamenetskii, in Encyclopedia of Condensed Matter Physics, 2005 B-DNA It (see Figure 4a) consists of two helically twisted sugar-phosphate backbones stuffed with base pairs of two types, AT and GC. 9 many different types of backbone modifications have been reported for sirnas. This backbone is composed of alternating sugar and phosphate groups, and defines directionality of the molecule. The sugar-phosphate backbone forms the structural framework of nucleic acids, including DNA and RNA. Hydrogen is used to construct the molecules water and organic compounds with carbon. Phosphate Backbone DNA and RNA, Biophysical Aspects M.D. the natural negatively charged phosphodiester backbone of rna is beset with poor cell-membrane permeability and is a substrate for ubiquitous ribonucleases, which rapidly degrade the rna to monomers and/or short oligomers. A sugar-phosphate backbone (alternating grey-dark grey) joins together nucleotides in a DNA sequence. In the structure below, each nucleotide is drawn in a different color. Carbon is also used to construct the energy-rich molecules adenosine triphosphate (ATP) and guanosine triphosphate (GTP). The backbone has a 5 end (with a free phosphate) and a 3 end (with a free OH group). Even the cell membranes are made of proteins. Carbon is an important element for all living organisms, as it is used to construct the basic building blocks of life, such as carbohydrates, lipids, and nucleic acids. These elements form the basic building blocks of the major macromolecules of life, including carbohydrates, lipids, nucleic acids and proteins. The sugar-phosphate backbone provides a ladder for the bases. The first four of these are the most important, as they are used to construct the molecules that are necessary to make up living cells. The sugar-phosphate backbone is very important in the structure of DNA for many reasons. They are carbon, hydrogen, nitrogen, oxygen, phosphorus, and sulfur. In biology, the elements of life are the essential building blocks that make up living things. Repeating unit in nucleotide polymer, with alternating sugar-phosphate backbone and one phosphodiester bond/linkage per unit/nucleotide residue (the. 
Covalent bonding involves the sharing of electrons between atoms, unlike ionic bonding where an electron or pair of electrons are transferred from one atom. Two nucleotides, each with an internal phosphoester bond and with a new phosphoester bond connecting them (spanning 5' of one sugar to 3' of the next): The sugar phosphate backbone is held together by the covalent bonding present between one nucleotide and the phosphate of another nucleotide adjacent to the sugar of the initial nucleotide. Schematic of single nucelotide featuring vertical line as phosphoester bond (to 5' C): RNA is unique among biological macromolecules in that it can encode genetic information, serve as an abundant structural component of cells, and also possesses catalytic activity. Since the repeating unit contains two phosphoester bonds, and the phosphates alternate with the sugars in the sequence, we call this a sugar-phosphate backbone held together by phospho diester bonds/linkages. Structures of RNA RNA A polynucleotide consisting essentially of chains with a repeating backbone of phosphate and ribose units to which nitrogenous bases are attached. So in a polymer of multiple nucleotides (DNA, RNA), the repeating monomer unit contains two phosphoester bonds, on "top" (5') and "below" (3') the sugar. 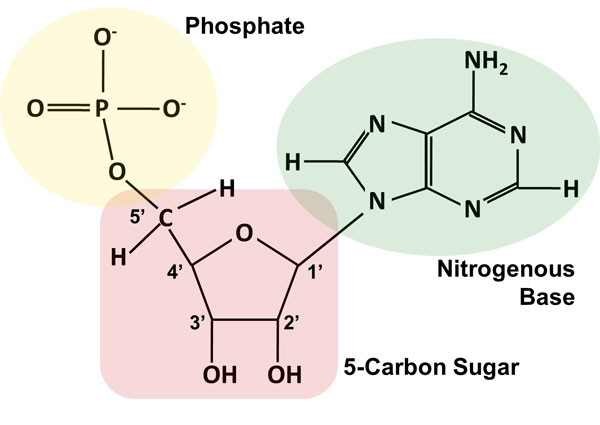
Additionally, two nucleotides are connected by one phosphoester bond (between a phosphate O and sugar 3'-C). Each nucleotide contains one phosphoester bond (between a phosphate O and sugar 5'-C).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
